Quality Collaboration by Design (QCBD™)
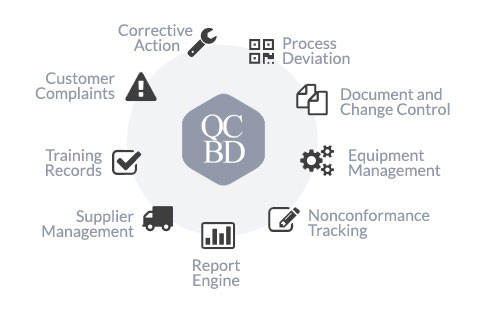
QCBD’s software suite is an affordable and fully integrated Quality Management Software for managing all your quality system data.
Designed specifically for manufacturing companies, QCBD
dramatically reduces the cost of achieving and maintaining compliance to quality management standards.
Manage Compliance
to
Quality Standards
ISO 9001
ISO 13485
AS9100
TS/IATF 16949
SQF 2000 / ISO 22000
Manage Standards ISO 9001, ISO 13485, AS9100, TS/IATF 16949 and SQF 2000 / ISO 22000.
SECURE, FAST & SCALEABLE
QCBD is a Windows-based client server application (not a web application) and is designed to be scalable, growing with your business. Start small and add modules and upgrade when you need it
GREAT VALUE
QCBD has the lowest total cost of ownership of any Quality Management Software (QMS) available – guaranteed.
FEATURE OVERVIEW
Modules contain specific functionality, but QCBD’s general features include:
 Email Notifications – Automatically alert users with tasks and distribute data records and reports.
Email Notifications – Automatically alert users with tasks and distribute data records and reports. Unlimited User Seats/Licenses – A single license supports your entire team. No individual user licenses necessary with QCBD.and reports.
Unlimited User Seats/Licenses – A single license supports your entire team. No individual user licenses necessary with QCBD.and reports. Dashboard View – Users can easily view and respond to all open action items.
Dashboard View – Users can easily view and respond to all open action items. Powerful Reporting – Select, group, and filter data in meaningful ways. Performance trending and Pareto analysis help you drive continuous improvement efforts with focus. Ability to export reports
Powerful Reporting – Select, group, and filter data in meaningful ways. Performance trending and Pareto analysis help you drive continuous improvement efforts with focus. Ability to export reports Electronic Signature – E-Signatures enable users to approve, route and store records online. Meets requirements for 21 CFR Part 11 (Electronic Records and Electronic Signature)
Electronic Signature – E-Signatures enable users to approve, route and store records online. Meets requirements for 21 CFR Part 11 (Electronic Records and Electronic Signature) User Permissions – User specific security, from read-only rights to full supervisor permission.
User Permissions – User specific security, from read-only rights to full supervisor permission. Attachments – Upload and attach digital photos, scanned images and documents to make them part of the record. Ability to check out / check in attachments.
Attachments – Upload and attach digital photos, scanned images and documents to make them part of the record. Ability to check out / check in attachments. Power Search – Find, sort, filter data to quickly find what you need.
Power Search – Find, sort, filter data to quickly find what you need. Customize – QCBD is highly configurable. Customize module screens and printouts to suit your organization’s terminology and descriptions.
Customize – QCBD is highly configurable. Customize module screens and printouts to suit your organization’s terminology and descriptions. Connect Remotely – Access your data from any location.
Connect Remotely – Access your data from any location.
FDA 21 CFR Part 11 compliant
Electronic records, electronic signature, audit trails, security
 Dual authenticated user ID with password
Dual authenticated user ID with password Full Audit Trails (date/time stamps of all events)
Full Audit Trails (date/time stamps of all events) Configurable password shelf life
Configurable password shelf life Notification of failed login attempts
Notification of failed login attempts User lockout after failed login
User lockout after failed login Configurable inactivity timer
Configurable inactivity timer Configurable record access restrictions
Configurable record access restrictions Software validation scripts available
Software validation scripts available
